Quality Management System
Radiant Distributions Limited, a leading distribution company with unmatched quality in the distribution arena, aims to deliver quality products through quality outcomes by execution of excellent strategies based on Quality Management System. The company operates in unified Quality Framework destined to regulate overall components within the framework and thereby enhances customer satisfaction.
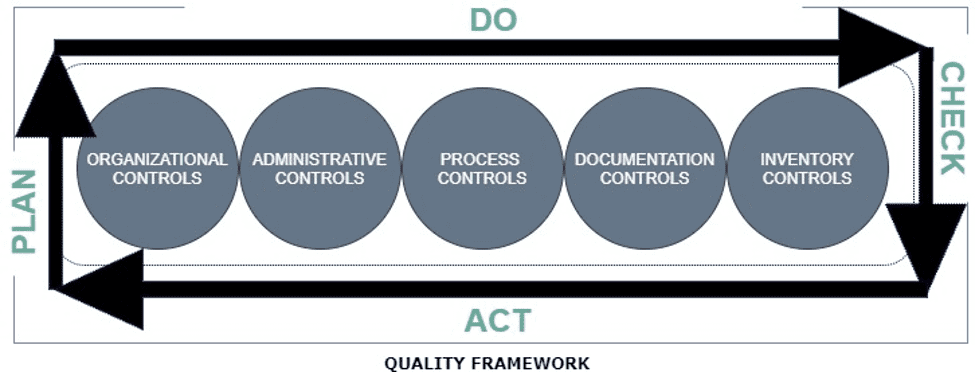
With quality systems compliant with WHO good distribution practices for pharmaceutical products, our quality compliance department has created a culture of Quality throughout the organization and strive to ensure that each and every individual churn out quality products and services to customers with highest level of confidence.
Radiant Distributions Limited functions independently from Operations. The roles and responsibilities of Quality Compliance department is to ensure that all processes comply with regulatory requirements. Our distribution process is integrated with periodic quality inspection assuring that all systems are thoroughly maintained and products delivered to customers are of the highest quality. In addition to that, the Quality Compliance department in conjunction with manufacturer provides accurate oversight of the release and / or rejection of imported finished pharmaceutical consignments with proper maintenance of documentations as per internal documentation management system. Any deviations relating to products / processes are handled as per established procedures and investigations are conducted to identify root cause and derive corrective actions and preventive actions appropriately. To evaluate quality systems, our distribution system is periodically audited by world-class pharmaceutical companies and recognized as the best in the territory.
Environmental monitoring in distribution chain is the key for determination of thermal hot spot and cold spot and foundation for acquiring knowledge about the state of the environment throughout the distribution chain. With advanced methodologies and tools that meet accreditation standards, a unique approach compliant with WHO good distribution practices for pharmaceutical products is applied to spot differences in temperature for identification of hot and cold spot. A comprehensive guideline is established and followed to go through the validation maze with proper documentation.
Radiant Distributions Limited remains solely committed to the quality management that will yield continual improvements thus creating a pathway to attract/retain new/existing business partners and customers.















